|
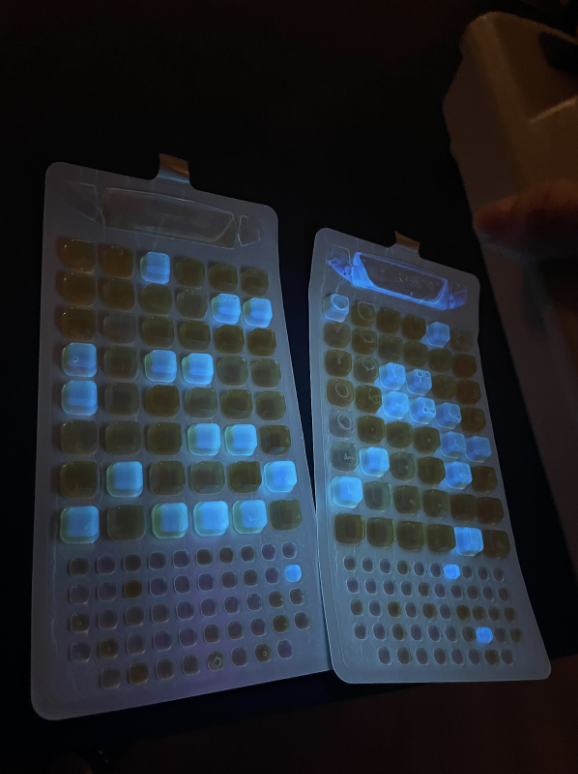
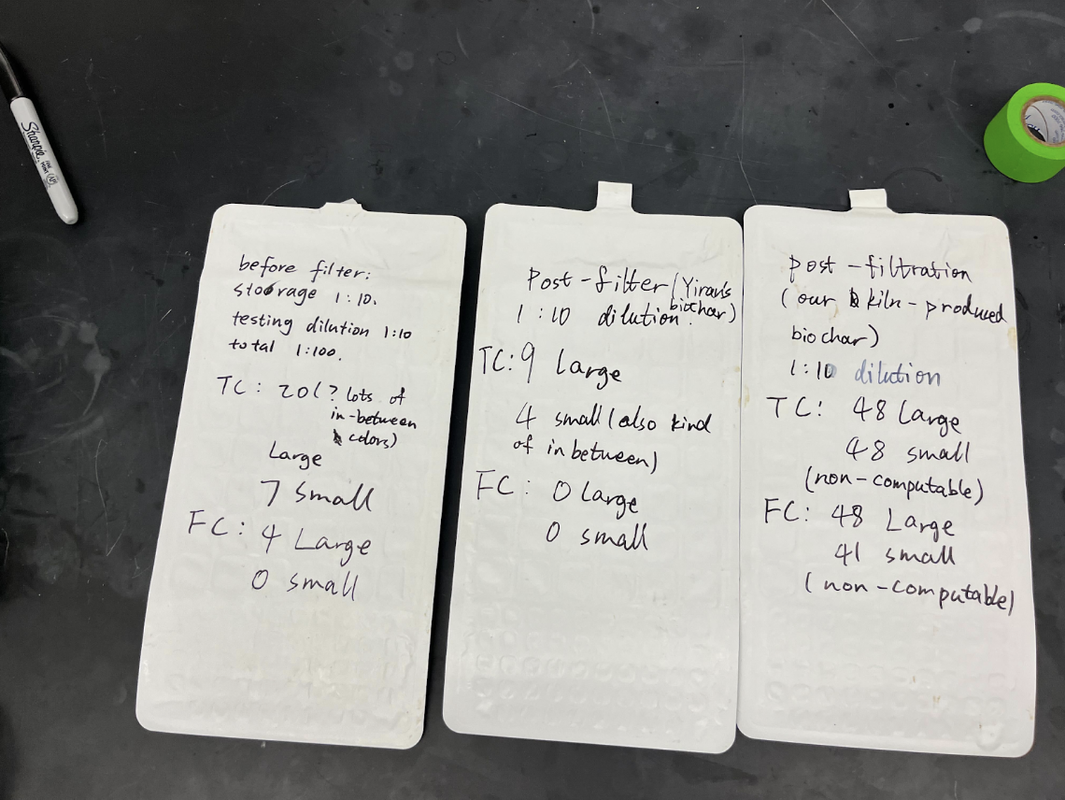
Now that we have our prototypes, we’ve spent the last few weeks testing them! We’ve experienced lots of challenges and setbacks, but they have helped us learn so much and improve our prototypes. Kiln: At this point, we’ve conducted four different kiln “burn trials”. We followed the protocol explained in the last post, if you are curious about the exact measurements we took and information we recorded. To give a quick overview, the way we test our kiln is to find bricks/rack to place the kiln on (to allow for airflow through the holes on the bottom), fill the barrel with feedstock (corn husks, rice husks, corn cobs) about halfway, use a lighter to light the feedstock, place the top/chimney on the barrel, and watch! When the burn looks like it’s going out (or when we want to stop the burn), we use a hose to dump water in the kiln until the fire is out. Throughout our entire testing process, safety is paramount, and we make sure to take safety precautions such as wearing eye protection and being cautious of hot temperatures. Trial 1: Our first trial was more of a proof-of-concept, and helped us get a handle on the process of lighting and putting out the kiln. We conducted the test at the Lake Lag fire pit, and used corn husks from the farm as our feedstock. The fire was really easy to light (which is something that last year’s team struggled with, however we quickly encountered a couple of major issues: first, the burn was extremely dirty and there was a ton of smoke, and second, the chimney fell off :(. We did get some char out of the first trial, however, and it was very fine. We thought the dirtiness of the burn might have been impacted by preparation of feedstock, since the corn husks are super light and pack a lot of air between them. This air could contribute to the dirtiness of the burn. This informed changes we made for our second trial. Trial 2: Our second trial was pretty similar to our first—we conducted the test at the Lake Lag fire pit, and also used corn husks. However, this time, we made sure to rip the corn husks up a lot more and push them down in the barrel to minimize air. We also reattached the chimney, using the same adhesive but giving it a lot longer to cure this time (one weekend), instead of testing 24 hours after attaching the chimney. We followed the same procedure as the first trial. Unfortunately, we also experienced the same issue of the burn being very dirty and producing lots of visible smoke. We decided to put out the kiln before it could finish pyrolysis, because it was producing so much smoke. One of the recurring questions that we’ve been facing in our burn trials is if we are actually performing pyrolysis, and if we are actually creating biochar or just charcoal. At the end of the day, the line between these two is so fine, especially for an imperfect (non-science-lab) kiln made out of repurposed materials. We have been using temperature and dirtiness-of-burn as indicators of pyrolysis (not just regular combustion), but this is imperfect and ultimately only lab-testing would be able to confirm, and this is something we will consider down the line. Now, our major priority is testing how our char/biochar works in a filter. Trial 3: Our third trial took place at the Stanford O’Donohue Farm. This was closer to where we were storing our project materials and obtaining feedstocks for our burns. Following proper safety procedures, we placed our kiln on four heavy concrete spacers atop a gravel patch, as well as wet the ground with a hose beforehand. By this time, we had drilled side holes into the top of our kiln. We wanted to see whether this would make a difference in the burn’s smokiness. Our burn was nonetheless smoky. However, one interesting insight was that when we removed the lid and chimney, the smoke became significantly clearer compared to before. This was a sign that insufficient airflow was likely a driving factor behind our dirty burn, rather than just the choice feedstock. We also observed a cleaner burn when testing the previous team’s smaller kiln prototype. It didn’t have a tall chimney, which allowed more air to reach the flame and reduced smokiness. In response to this, we quadrupled the amount of airholes on the kiln’s bottom face and tripled the number of side holes at the top. We hoped this would reduce smokiness for our fourth burn test. Trial 4: Our fourth trial saw another corn husk burn, this time with more air holes. By this point, we had become familiar with the experimental design of a burn test, which made the procedure go very smoothly. The test was brief. Unfortunately, the resulting smoke from this burn was still opaque and dirty, meaning we had not added enough airholes to make a significant difference. The chimney also fell off during this burn test (the third time it did this across our trials), implying we needed to find a more robust means of attaching it. Rather than add even more additional airholes the next time, we decided to test our next burn with a heftier feedstock to see if that would make an unforeseen difference. Trial 5: On our fifth and most recent trial, we switched the burn’s feedstock to corn cobs instead of corn husks. Cobs had exponentially more mass and rigidity in their shape than husks. We filled the kiln with about 20 corn cobs, then interspersed husks between the top layers to act as kindling. We then began the burn by lighting one husk on fire, and dropping it into the middle of the kiln. In general, this burn brought some better news. It lasted 10 minutes, since we allowed it to go on for longer than previous ones. During the burn, temperatures reached 500 degrees Fahrenheit across all parts of the kiln, even peaking at 600 towards the top of it. The burn became less smoky as it went on, which was desired. A reduction of smokiness over time was something we observed in videos of successful kilns online, so this could be seen as a sign we were moving in the right direction. This could have also been due to the fact that we conducted this test without a soup can chimney, since we did not have time to attach it beforehand. This likely increased airflow to the feedstock, cleaning up the burn. It was unclear to tell when to stop the burn, which is a question we should research the answer to. We quenched it when solid particulates began to rise out of the chimney. Afterward, we observed that our corn cobs had blackened completely and shrunken significantly in size. They were also much easier to snap in half than they were at the beginning - a sign that we changed the material in a promising way. Quality biochar is porous, which translates to less structural integrity. If it’s porous, it means it may be effective at filtering water. What now? As it stands now, our kiln continues to face the issue of smoky burns and insufficient temperatures for pyrolysis. Our immediate goal is to talk with experts on kiln thermodynamics and biochar production so we can better understand why our models are performing the ways they are. We also plan to test our current TLUD design once more with corn cobs using a soup can chimney, to see if it affects airflow and smoke clarity, as well as with rice husks, to see if a tightly compacted, airtight fuel makes a difference in amounts of smoke produced. We also have ideas in mind for modifying our prototype. These include inserting an enclosed metal container filled with feedstock into the center of our kiln, then burning additional feedstock around the enclosed container to bake its contents. This would effectively convert our kiln into a retort. Finally, we may modify a paint can to serve as a second barrel in our TLUD, where we’d fill it with feedstock, insert it into our current kiln, and light it. This may help insulate the feedstock and emulate other conventional two-chamber TLUD designs. Filter: Trial 1: The test result for the filter part varies greatly from our initial expectations. We conducted the first trial during week 7, using our prototype with the PVC pipe attached on the side, the corn husk biochar created from our kiln, fine sand, and small pebbles. After 24 hours of incubation, we got the following result: In the pictures above, the tray on the right contains our pre-filtered stock water, while the tray on the left contains water after filtration, both diluted by 1:100. As mentioned in our previous blog post, the pre-filtered stock water is a mix of Lake Lagunita water, Codiga waste water, and tap water. The luminescent boxes indicate presence of E. coli, while yellow boxes indicate the presence of total coliforms. As the picture illustrates, our filter didn’t properly carry out its function of removing harmful bacteria. The difference in total coliform and fecal coliform levels are not significantly altered. Upon reflection, we proposed that there could be three reasons behind our initial failure: 1. The end product of our kiln did not meet biochar standards 2. Biochar alone doesn’t have the ability to effectively remove E. coli 3. There’s a problem in the design of our prototype In order to deduce the reason behind this failure, we did our second test in parallel with a control group, using our stacked bucket prototype and commercial biochar kindly provided to us by Yiran Li, a PhD student at the Luthy Lab. The commercial biochar is from douglas fir and is industrial grade, so we would expect that it would remove E. coli from the water samples. Test result can be seen here: This picture contains the numerical result of the color of indicators, same as the first test, with TC indicating total coliform, FC indicating fecal coliform, large and small indicating the number of large and small chambers that indicates presence.
While comparing the numerical result with the IDEXX reference sheet, we learned that there are around 400 MPN/100mL E. coli in the stock solution prior to filtration, 0 in the water after filtration through the second prototype (stacked bucket model using commercial biochar), and more than 24000 MPN/100 mL E coli. in the water after the second trial of filtration through the first prototype (side-outlet using kiln-produced biochar). The conclusions we can draw from these results are that: 1. High quality commercial biochar does have the ability to remove E. coli, 2. E. coli actually proliferate in our first prototype, contaminating the water instead of filtering it, suggesting that there is a flaw in our design After consulting Yiran, we learned that the growth in E. coli is not uncommon, because the bacteria feeds on dissolved organic carbon, which is abundant in the biomass, such as corn husk, that we used to produce biochar. Yiran mentioned that is will be difficult to filter significant amounts of E. coli out of our water sample without using chlorination or other more advanced methods. In the next few weeks, we will reevaluate how plausible it is for us to continue focusing on the complete removal of E. coli. It may be more realistic to focus on the removal of heavy metals instead. Thank you so much for reading our post! In conclusion, although we've encountered many challenges, we've learned so much from them and are better designers and engineers because of it!
0 Comments
Now that we have finished the first version of prototypes for both the kiln and the filter, our next step is to conduct testing! Testing is vital because it helps us understand the effectiveness of our prototypes, and also helps us narrow down which aspects of our prototypes need to be improved or changed for the final version. Kiln: On the kiln side, our testing mainly revolves around calculating the yield rate of the kiln, as well as determining if the char produced from the kiln is actually biochar—-meaning that it conforms to biochar standards including pH, pore size, and more. Additionally, our testing also helps us better understand the thermodynamic properties of the kiln, as well as the “user experience” of our kiln. To summarize, the big picture testing plan is:
When conducting the burning trials, we have a detailed list of information and observations to collect that include initial and final weight of biomass, temperature measurements during the burning, and more. The most detailed version can be found in the “Kiln Test Data Recording Sheet”, which is what we have been using to record our data. All of the testing and data we collect tie back to our core user and technical requirements. First of all, it’s vital to measure and calculate the yield percentage (mass of biochar out over mass of biomass in), in order to understand the efficiency of our kiln. This is important to ensure that our users are getting the most out of their investment of time and effort in using the kiln. The next major requirement is that the biochar actually meets international biochar standards, and is effective as a water filtration material. This is why it’s so important to test the biochar itself, as well as monitor the burning process for indications that pyrolysis (the process that produces biochar) is occurring instead of just traditional combustion. These signs that help us distinguish between pyrolysis and traditional combustion include: temperature, flame color, and if the burn is dirty or not dirty. Another core requirement is safety, which ties into many of the protocols we have been following and data we have been collecting. For example, the temperature of the kiln at different times during the burn and the water required to put out the kiln are all things that will inform the safety precautions that must be followed by our users when using the kiln. To give an update, we have already conducted two kiln trials. In the first trial we encountered many problems, mainly that the chimney fell off the kiln itself and that the top piece of the kiln was unstable. In response to our findings, we reattached the chimney, and adjusted the design of the kiln to make the top more stable. Next, we conducted our second trial. Despite these changes, we still encountered a very fast and dirty burn, which is an indication that pyrolysis might not be actually occurring. In our next we plan to test a different biomass than corn husk to see if the corn husks are the problem, as well as meet with a thermodynamics professor to give us some more hands-on advice on our kiln design. Filter: As far as the filter side goes, our team has been working alongside Dr. Kopperud in developing a testing plan that quantitatively measures the effectiveness of our filter at removing coliforms and E. coli as well as the lifetime of our biochar filter. After consulting with Dr. Royal Kopperud and his graduate students, we became aware that removing chloride and fluoride ions would be quite difficult since these ions penetrate through carbon filters and would require more advanced methods such as reverse osmosis and distillation. In the past few weeks, we have been working to develop a water sample that is representative of the water quality in northern Malawi. By diluting wastewater from Codiga by a factor of 100, we were able to develop a water sample that is representative of the E. coli and coliform concentrations (1850 MPN/100ml), as stated in a research paper by Jade Ward (1000-2000 MPN/100ml). After creating a representative water sample, we conducted an IDEXX test to determine the concentrations of coliforms and E. coli. The yellow boxes below indicate the presence of E. coli while the fluorescent boxes indicate the presence of coliforms. Here is our testing plan for the coming weeks:
We are looking forward to continuing the testing process, and learning from and improving our prototypes! Thanks for reading! Welcome to our first blog post of 2023! The new Biochar team has been hard at work developing and revamping previous prototypes from previous teams. In this blog post, we’ll update you on the progress we’ve made in the past few weeks and highlight the key insights we’ve learned from the prototyping process thus far. Filter: Last quarter, the Biochar team pivoted away from the multi-bucket filtration system and started focusing on sizing down the filtration system to 2 buckets. The new model features a smaller bucket that sits atop a larger plastic bucket. After building the new prototype last quarter, the objective this quarter is mainly testing and perfecting this prototype. In the first few weeks of this quarter, we carried out some preliminary mechanical tests for the filter, including stability, filtration speed and leak-proof features. We encountered some challenges, such as biochar tainting the water black because we did not add a filtration cloth at the water outlet, and slow filtration due to mistakenly using water-absorbing polymeric sand in the filter. However, we learned from these setbacks and made necessary adjustments to mitigate the issues. For the remainder of the quarter, we plan to carry out extensive water quality tests, with a focus on coliform and E.coli. We reached out to Professor Royal Kopperud, who generously agreed to provide us with lab space and IDEXX E.coli/coliform testing equipment. With his support, we hope to optimize the functions of our filter in removing microbes and satisfying the demands of our target communities. We're excited to continue testing and improving our prototype for the rest of spring, as each step brings us closer to achieving our goal of providing safe and clean drinking water. Kiln: This quarter, on the kiln side of the project, we are focused on building a larger kiln prototype. Based on the feedback we’ve received from our community partner, WESM, we decided on a size of around 5 gallons. The goal of building the larger prototype is to get a more accurate sense of the yield, burn time, and general thermodynamic behavior of a larger size kiln, since it differs significantly from that of a small kiln like the ones previously prototyped. Furthermore, building a larger kiln will hopefully allow us to actually use our biochar in the water filter (as opposed to store-bought biochar). The first step of this process was sourcing materials, which actually ended up being harder than expected. For the main body of the kiln, we started by ordering a 6 gallon mini trash can. However, when it arrived, we learned that it was made of galvanized steel, which is steel coated in zinc. We learned from our research that galvanized steel releases zinc fumes when heated, which are extremely toxic to breathe. As a result, we decided to find a different metal bucket to use. After checking lots of websites, calling companies, and reading reviews, we finally found a metal bucket that wasn’t made of galvanized steel, and also didn’t have an epoxy coating (common of many larger metal containers). Next, we started the construction process, which we are still in the middle of. Following the guidance of an online tutorial and several different Youtube videos, we started by adding air-flow holes to the bottom of the main container. In the PRL, we used a ⅜ bit to drill a 3x3 grid of holes in the bottom. Our next major task is to work on the top chimney part of the kiln. This involves cutting a large hole in the lid of the main can, so that the chimney can sit on top. To make the chimney, we are planning on stacking several soup cans on top of each other (with their tops and bottoms removed).
We look forward to finishing the construction process and being able to start testing! Thanks for reading this post! |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |














 RSS Feed
RSS Feed
