|
Here, we detail the results of our tests, including what went well, what went wrong, and our general reflections.
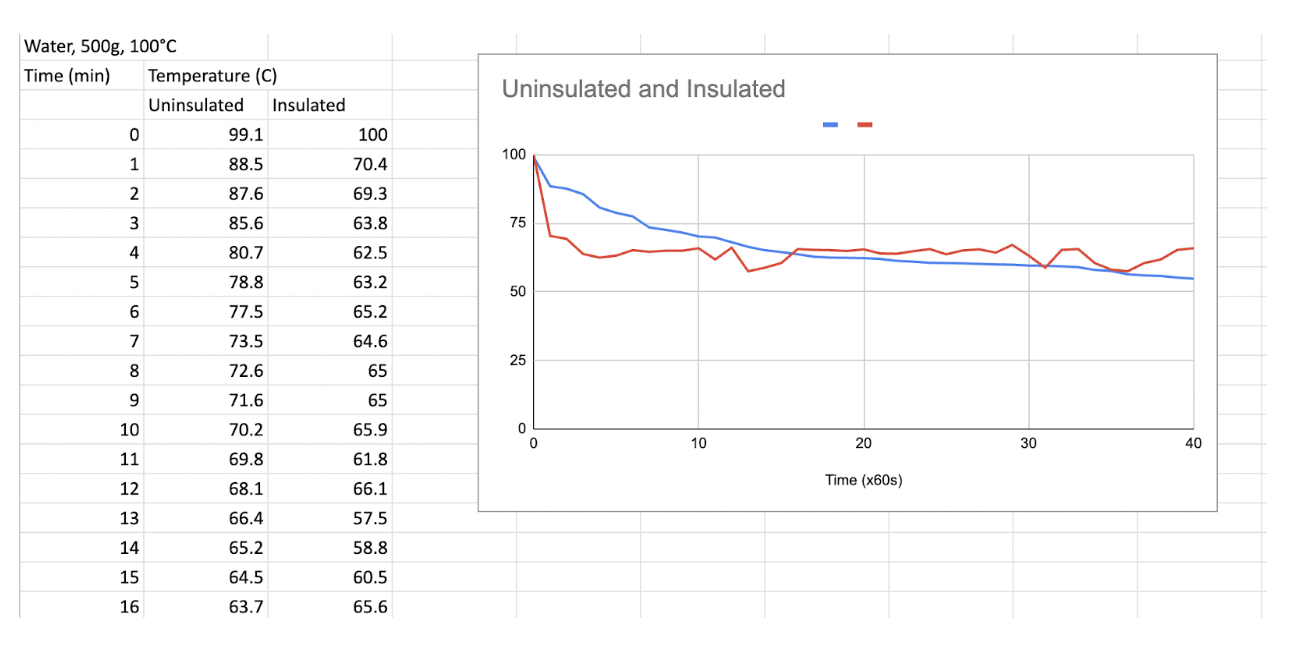
The results were not as accurate as we had hoped, mainly because the thermometers for the insulated test demonstrated a quicker drop in temperature than the uninsulated test. However, after analyzing the data further and constructing graphs, we quickly realized that the insulated test retained the temperature for longer at a steadier rate, compared to the uninsulated. Moreover, the insulated test was clearly far warmer, with steam rising from the device for more than an hour. Contrastingly, the uninsulated pot did not display the same level of evidence of heat. As for future tests, we can definitely ensure that the thermocouple is first properly aligned and tested accurately by conducting placebo tests on different substances with known temperatures, then comparing to see if they are the same.
0 Comments
The first iteration of our prototype is focused on the Thermal Energy Storage (TES) aspect of the product. This follows from our user and technical requirements that we want to create a cookstove that’s able to adequately replace current methods of cooking, which means that it has to be usable at all times of the day and be compatible with the region's electricity infrastructure. Concretely, from a technical standpoint, it has to be capable of operating with off-grid solar and grid infrastructure, and from a user standpoint, it has to operate at least 300 degrees Celsius. As such, our testing plan’s main objective is to determine the efficiency of the thermal energy storage prototype. Below is a testing plan we plan to use for our first major test – testing insulation. We will follow this same structure for future tests.
Comprehensive Testing Plan Objective: To determine the efficiency of the Thermal Energy storage prototype in terms of the rate of heat loss and how much it can be minimized, how long Materials and Equipment:
Procedure: Testing Effect of Insulation:
Variables
Data Analysis
Predicted Results Q = m x c x t = (0.5 kg) (4.18 kJ/kg x c) (100 - 17.2 degrees C) = 173 kJ heat loss total Rate: Newton's Law of Cooling: t = -ln[ (T - Tambient) / (Tinitial - Tambient) ] / k
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
